Of all best practices for the storage and handling of pharmaceuticals, perhaps none is more crucial than temperature monitoring. As complex chemical and biological products, medications can easily be rendered impotent – even toxic – when subject to excursions outside of their prescribed temperature range. Even the simplest products are rarely unaffected by extremes of heat or cold.
The safety of some pharmaceuticals in particular is at risk if exposed to improper temperature variations. For instance, overheated products promote the growth of pathogens, while freezing reduces the efficacy of medications containing proteins. Public health can be endangered if medications lose their strength, especially vaccines, which leave people exposed to life-threatening infectious diseases. Unfortunately, temperature monitoring errors result in expensive revaccinations of many patients and significant financial loss due to wasted vaccines.
Because federal regulatory agencies recognize the many costs of temperature excursions, traceability by means of a recorded temperature history is now required as a mandatory component of GDP compliance. Nevertheless, temperature monitoring practices used in many safety plans are outdated: intermittent readings, manual data entry, paper reporting, and inefficient sharing methods. The result? Inconsistencies, data gaps, and delays that can undermine the temperature surveillance process.
Digital Temperature Monitoring Devices (TMDs) Have Come of Age
Decades ago, pharmacies had no choice but to make do with standard mercury or alcohol thermometers, the only measuring technologies readily available. Any system dependent on these devices was, therefore, limited to periodic manual inspection and recording of single-point temperature measurements. While so-called “max-min” thermometers could indicate the lowest and highest temperatures reached, they could not pinpoint the time of occurrence.
By the turn of the 21st century, many drivers put pressure on the pharmaceutical industry to begin using automatic digital systems:
- Increased regulatory oversight
- Growth in biotech and bio-generic products
- Introduction of direct-to-patient strategies
- Technological innovations such as wireless communications and cloud storage
- New supply chain integration models for improved data transparency
- Investment in global cold chain infrastructure
The earliest automatic systems used battery-powered temperature recorders capable of remote, continuous temperature measurement and data storage on an external device for subsequent analysis and distribution. Although this new functionality makes checking temperature easier, such systems remained too dependent on inefficient manpower resources, time, and training – all of which compromise the frequency and magnitude of reliable monitoring. For this very reason, organizations such as WHO and CDC now strongly recommend automatic, remote, continuous temperature recording synched directly to a digital data logger.
Anatomy of a Digital Data Logger (DDL)
In the most basic sense, a “data logger” is any computer-based device that can be used to store statistics and information, which could include plug-in boards, serial communications, or wireless transmitters to a mobile device. But those configurations are already superseded. Most instrument manufacturers today define a “digital data logger” or DDL as a stand-alone device that can read various types of electrical signals and store the data in internal memory for later download and analysis independent of a computer.
In the most recent Vaccine Storage and Handling Toolkit, The Centers for Disease Control and Prevention (CDC) now specifically recommends the use of digital data loggers with the following features:
- Detachable probe that best reflects vaccine temperatures (e.g., a probe buffered with glycol, glass beads, sand, or Teflon)
- Alarm for out-of-range temperatures
- Low-battery indicator
- Current, minimum, and maximum temperature display
- Recommended uncertainty of +/-0 .5° C (+/-1° F)
- Logging interval that can be programmed by the user to measure and record temperatures at least every 30 minutes
Located inside each refrigerator and freezer unit along the pharmacy cold chain, DDLs best provide the detailed reports into temperature variations that enable accurate analysis of product potency. Unlike a thermometer that shows only the coldest and warmest temperatures reached in a unit, a DDL provides information about temperatures recorded at intervals that can be preset using web-based software. The most accurate DDLs use a buffered temperature probe that measures the actual temperature of the vaccines, whereas standard thermometers record ambient air temperatures, which can vary widely throughout a storage unit.
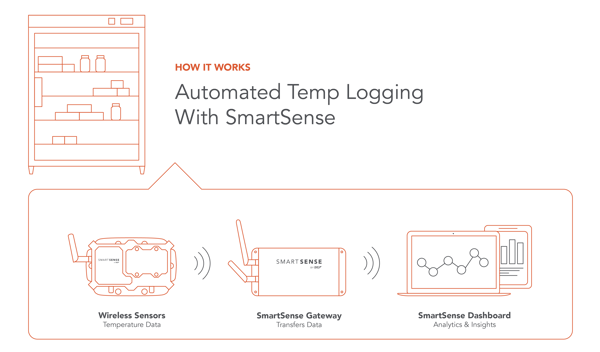 Digital Data Logging with SmartSense.
Digital Data Logging with SmartSense.
Although a DDL is not yet mandated as the required monitoring system, the CDC specifically warns against the use of the following temperature monitoring devices (TMDs), which compared to a DDL, may fail to detect a significant temperature excursion:
- Alcohol or mercury thermometers, even if placed in a fluid-filled, biosafe, liquid vial
- Bimetal stem TMDs
- TMDs used for food
- Chart recorders
- Infrared TMDs
Finally, a DDL is only as good as its Certificate of Calibration. To ensure the accuracy of its readings against nationally accepted standards, calibration testing should be conducted annually or according to the manufacturer’s suggestion. That’s because all TMDs can “drift” over time with constant use. However, don’t wait if you think a DDL may have been physically damaged – get a replacement ASAP.
The Silver Lining of a Digital Cloud
DDLs that store data to an external hard drive may require onsite analysis, delaying a proactive correction of temperature excursions that may be ultimately too late. Instead, the combination of new-generation devices and the latest data transmission, capture, sharing and analysis technologies implemented on a digital cloud is the latest and greatest development in temperature monitoring.
The problem with many DDLs is that they must be connected to a host device in order to download recorded data, which means that, in practice, they have no real-time data interactivity. With a cloud-based monitoring system, however, as soon as temperatures reach unsafe levels, alerts can be rapidly shared with key stakeholders – including pharmacists and managers who can take action to save the inventory. A cloud-based solution lets you focus on predicting and eliminating cold-chain breaks and temperature excursions rather than detecting and removing violated products from the supply chain.
Now Is the Time for a DDL
Real-time temperature monitoring is no longer “the future” – it’s right now. Today’s and tomorrow’s pharmaceutical supply chain is all about acting, not reacting. Cloud-based DDLs provide continuous live recording, automatic alerts, 24/7 data access, simultaneous data sharing, and proof of regulatory compliance. They offer the difference between instant and post-event notification of days or even weeks, which translates into the difference between a safe shipment delivery or millions of dollars of product waste, a regulatory investigation, and most important of all, failed patient care.
Subscribe to Connected Insights!
Subscribe to our blog to get regular email updates on food safety, pharmacy safety, and supply chain insights.

